|
December
2018 |
|
| How many contact angles are there? | |
| This month we will attempt to
identify and distinguish between a number of different types of
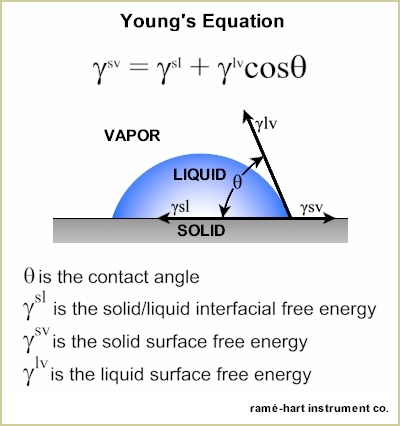
contact angles. Young Contact Angle. The Young equation shown below quantifies the energy that is found between the three phases - liquid (l), solid (s), and (v) vapor or air - that are present when a drop of liquid, deposited on a solid surface, reaches equilibrium.
The Young contact angle works on an ideal surface which is described by Bormashenko as a substrate that is atomically flat, chemically homogeneous, isotropic, insoluble, non-reactive and non-stretched.1 In the real world, very few surfaces can be considered "ideal". Observed Contact Angle. Since most surfaces have some degree of roughness or texture or contamination2, the observed contact angle reflects the local wetting behavior which can be described by the Young-Dupré equation. This equation introduces a spreading parameter (S) which is described in this equation: S = glv (cosθ - 1) When S > 0, the drop will completely wet (contact angle is 0°) and when S < 0, then partial wetting occurs. Contact Angle Hysteresis. As noted, most surfaces are not ideal. The deviations from perfect smoothness and topographical and chemical homogeneity result in a range of possible contact angles. This range is identified as the contact angle hysteresis and represents the difference between the maximum and minimum possible contact angles.
If the contact angle hysteresis is small, this indicates that the solid is quite homogenous relative to chemical and structural composition. Advancing Contact Angle. This is the maximum possible contact angle, or θa. When using the add/remove volume method (e.g., with our Automated Dispensing System3), additional liquid is slowly added to the drop. The drop will increase in volume and the contact angle will also increase until the three-phase line eventually slips. Prior to the slip, the maximum, or advancing contact angle is captured. When using the tilting base method4 (e.g., with our Automated Tilting Base), the downhill side of the drop represents the advancing contact angle as shown in the graphic below.
Receding Contact Angle. The counterpart to the advancing contact angle is the receding contact angle, or θr - the minimum possible contact angle. Using the add/remove volume method, we retract volume slowly until the three-phase line contracts. We capture the minimum possible contact angle just prior to the dewetting. With the tilting base method, it's the contact angle on the uphill side of the drop - just prior to roll-off. Static Contact Angle. This often refers to the observed contact angle immediately after the drop is deposited. If the surface is ideal and the external phase is sufficiently saturated, then a drop of pure liquid would [theoretically] remain static in perpetuity. Dynamic Contact Angle. On real world surfaces, however, wetting, evaporation, or a change in temperature or other environmental condition can result in a change of contact angle over time. This is referred to as dynamic or time-dependent contact angle.5 Captive Bubble Contact Angle or an inverted sessile drop is measured when the external phase is a liquid and the drop phase is a gas which is deposited on the underside of a substrate as shown in the picture below.6 This type of measurement is very useful on absorbent and superhydrophilic solids which are difficult to measure using the conventional sessile drop method.
Equilibrium Contact Angle. See observed contact angle above. The equilibrium, or stable, or apparent contact angle will be somewhere between the advancing and receding contact angle. Water Contact Angle. Since water is the most common liquid used to measure contact angle, it's the water contact angle that determines if a surface is hydrophobic (water contact angle greater than 90°) or hydrophilic (water contact angle less than 90°). If the test liquid is oil and the contact angle is high, then the surface is said to be oleophobic or lipophobic. If a surface has a high contact angle for all (or most) liquids, it's said to be omniphobic. The converse behavior is oleophilicity and omniphilicity. There are, by the way, surfaces that are simultaneously superhydrophobic and oleophilic.7 In short, there are a variety of contact angles. Whether you need to measure equilibrium contact angles, dynamic contact angles, or the advancing and receding contact angles of a given solid/liquid system, we have a tool for you. Check out our current family of models here.
1 Bormashenko, Edward Yu.,
Physics of Wetting, De Gruyter 2017, Appendix 2A. |
|
| New Book: Physics of Wetting | |
|
Of interest to surface scientists is a
new book out titled Physics of Wetting: Phenomena and
Applications of Fluids on Surfaces by Edward Yu. Bormashenko
published by De Gruyter. Check out the preview
here. |
|
| Happy Holidays | |
|
Wishing you a joyous holiday season
and Happy New Year from all of us here at ramé-hart instrument co.
Thank you for your continued business. Without it, we would not be
the world leader in contact angle goniometry. |
|
|
Regards,
Carl Clegg |
|